Author: Megan Harper, Applications Scientist
Solving a Global Problem

Gardening is one of the most rewarding hobbies in today’s society. The process of planting from seed, watering each day, and watching a beautiful plant develop over time is rewarding, and even more gratifying when it is edible. The recent pandemic has resulted in an increased interest in gardening, as many took on the hobby to uplift their mood and get out of the house. This fascination with plants has also grown due to the Earth’s growing population and the resulting need for increased food production. Unfortunately, only so much can be done to a plant to increase its production, and often farmers are forced to use chemical options that pose a risk to overall food safety. Access to natural resources that promote the growth and development of crops, like phosphate, are diminishing due to the growing food supply chain (1). The big question is how can these problems be tackled if the resources and means to address the situation are unattainable? Fortunately, a part of the answer is rooted in plant volatiles.
Volatile Organic Compounds (VOCs)
Thousands of plant varieties exist, and of these, each possesses thousands of chemicals compounds that make up a diverse and unique plant volatile profile. These volatile organic compounds (VOCs) are comprised of mostly terpenoids, fatty acids, aromatics, and amino acid derivatives (1). Plant volatile determination has become popular as there is a desire for food sustainability, postharvest protection, and increases in crop yields to meet the needs of a growing population and support greener methods. VOCs found in all plant types can attract pollinators, induce allelopathic effects, and promote the growth and development of the plant itself while also inhibiting pathogens (2,3). Therefore, understanding the plant volatile profile can provide useful information to help increase crop production.
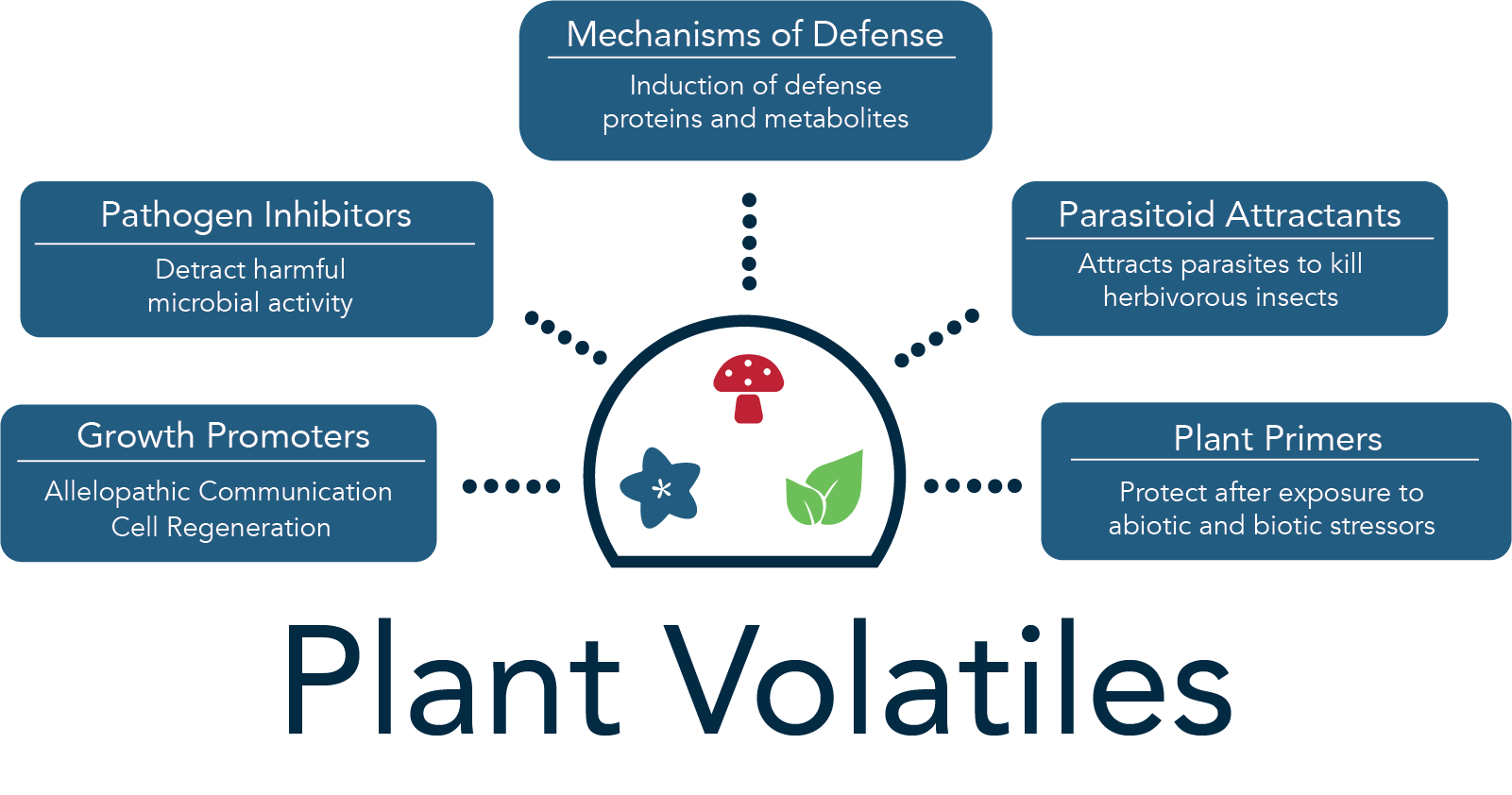
Plant Volatile Extraction Techniques
Active sampling with sorbent traps used in tandem with air sampling devices or dynamic headspace (DHS) sampling in a chamber are all established techniques used for extracting plant volatiles (2). However, these techniques have some disadvantages such as requiring the plant to be taken from its natural environment, requiring optimization of sorbent material, and the purchase of additional equipment (flow meters and sampling pumps). Passive sampling of volatiles in the field has been demonstrated using the GERSTEL Twister (3,4,5). This technique offers simplicity compared to the previous methods used for plant volatiles.
Obtaining a comprehensive list of all the volatiles emitted from every existing plant will be an arduous task, but GERSTEL sample preparation and automation technology can provide researchers with the tools they need to get started. The GERSTEL Twister, TF-SPME membranes, sorbent filled thermal desorption tubes, and Thermal Desorption Unit (TDU), in combination with GC-MS analysis, supplies analysts with the best tools necessary to acquire a detailed overview of plant emissions.
TF-SPME Method: A Case Study
 Using a polydimethylsiloxane/divinylbenzene (PDMS/DVB) TF-SPME device has shown to be a simple, sensitive, and thorough method for sampling plant volatiles (GERSTEL AppNote 240). In comparison to active air sampling with Tenax® TA and PDMS foam filled thermal desorption tubes, the TF-SPME device produces lower detection limits and better selectivity.
Using a polydimethylsiloxane/divinylbenzene (PDMS/DVB) TF-SPME device has shown to be a simple, sensitive, and thorough method for sampling plant volatiles (GERSTEL AppNote 240). In comparison to active air sampling with Tenax® TA and PDMS foam filled thermal desorption tubes, the TF-SPME device produces lower detection limits and better selectivity.
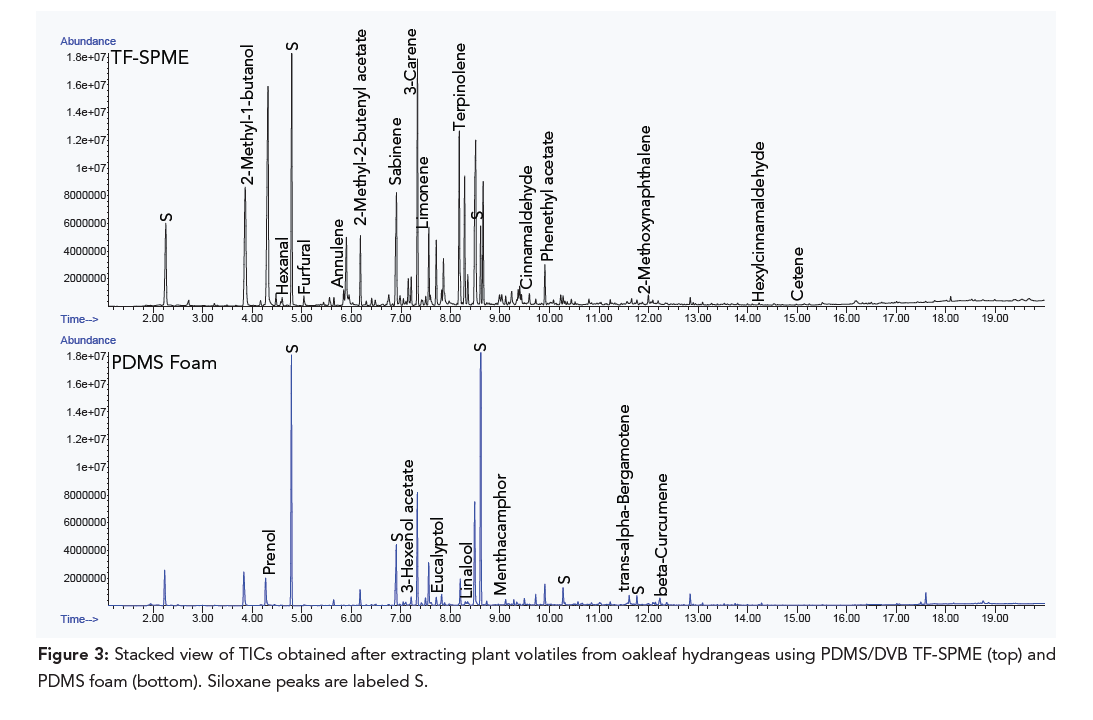
The time of day during which plant volatiles are sampled is also important. Plants follow an endogenous circadian rhythm to prompt volatile emission to attract pollinators (6). When analyzing oakleaf hydrangeas, overnight sampling with a PDMS/DVB TF-SPME device did not detect eucalyptol. However, when the oakleaf hydrangeas were sampled during the day using Tenax® TA thermal desorption tubes, eucalyptol was detected. Eucalyptol’s fresh, camphor-like smell is well known to attract bees.
The PDMS/DVB TF-SPME devices were shown to provide the best results for the plants used in this study. Passive sampling generally requires longer sampling times but can provide valuable plant volatile information without the need for sampling pumps. Among violet star petunias, oakleaf hydrangeas, citronella, and lemon thyme plants, the device extracted several volatiles that play key roles in plant communication, pollinator attraction, and defense. These included limonene, creosol, vanillin, trans-isoeugenol, benzophenone, eucalyptol, camphor, linalool, cinnamaldehyde, sabinene, geraniol, citronellol, and thymol.
The potential of using TF-SPME devices for plant volatile determination was shown by identifying several key plant volatile contributors. Active sampling using sorbents tubes can also be a useful approach for analyzing plant volatiles.
Conclusion
As researchers continue to acquire more information about plant emissions, GERSTEL’s sampling and analysis options will provide them with a simple, green, and comprehensive approach for detecting plant volatiles.
References
- https://web.mit.edu/12.000/www/m2016/finalwebsite/solutions/phosphorus.html
- Tholl D, Hossain O, Weinhold A, Röse U.S.R. and Wei Q. Trends and applications in plant volatile sampling and analysis. Plant J, 2021;106:314-25.
- Askenov, A, Pasamontes, A, Peirano, D, Zhao, W, Dandekar, A, and Fiehn, O. Analytical Chemistry, 86, 2481-88.
- Cheung, W, Pasamontes, A, Peirano, D, Zhao, W, Grafto-Cardwell, E, Kapaun, T. Metabolomics, 11, 1514-1525
- Kfoury N, Scott E, Orians C, and Robbat A. Journal of Agricultural and Food Chemistry.2017;65 (38): 8501-9.
- Baldwin IT. Plant volatiles. Curr Biol. 2010 May 11;20(9):R392-7.
